GROUNDWATER AND LEACHATE: EX-SITU CHEMICAL/PHYSICAL TREATMENT
Separation
Separation techniques concentrate contaminated waste water (through physical and chemical means) & detach the contaminants from the groundwater.
These (ex situ) separation processes are used mainly as a pre or post-treatment process.
- Apply to aqueous waste streams (ground water, lagoons, leachate, industrial waste-streams etc)
- Target contaminant groups: VOCs, SVOCs, pesticides, and suspended particles.
Note: Also solvents may be recovered for reuse.
Ex situ separation occurs using many processes (explained below):
1) Distillation
2) Filtration/ultra-filtration/micro-filtration
3) Freeze crystallization
4) Membrane Pervaporation/Reverse osmosis.
These (ex situ) separation processes are used mainly as a pre or post-treatment process.
- Apply to aqueous waste streams (ground water, lagoons, leachate, industrial waste-streams etc)
- Target contaminant groups: VOCs, SVOCs, pesticides, and suspended particles.
Note: Also solvents may be recovered for reuse.
Ex situ separation occurs using many processes (explained below):
1) Distillation
2) Filtration/ultra-filtration/micro-filtration
3) Freeze crystallization
4) Membrane Pervaporation/Reverse osmosis.
1) Distillation
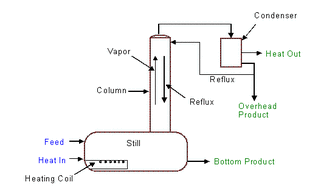
A Typical Distillation Unit
Distillation is a chemical separations process involving vaporization and condensation to separate components of varying vapour pressures (volatilities) in a liquid or gas stream.
Note: the mixture remaining in the still is enriched with the less volatile components (the bottom product).
- Generally successful separation of volatile organic components.
- Heat is applied to a liquid mixture in a still, causing a portion of the liquid to vapourise.
- The vapours are subsequently cooled and condensed - a liquid (distillate/overhead) is produced.
- The distillate is enriched with the higher volatility components.
Note: the mixture remaining in the still is enriched with the less volatile components (the bottom product).
- Generally successful separation of volatile organic components.
2) Filtration/Ultra-filtration/Micro-filtration
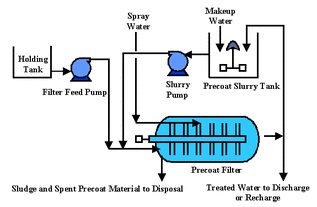
A Typical Filtration Unit
Filtration is the physical process of separation - based on particle size
Note: Ultra-filtration/ micro-filtration differs by forcing the fluid through a semipermeable membrane where only the particles smaller than the membrane openings flow through.
- Particles suspended in a fluid are separated by forcing the fluid through a porous medium.
- The suspended particles are trapped on the surface/within the body of the medium.
Note: Ultra-filtration/ micro-filtration differs by forcing the fluid through a semipermeable membrane where only the particles smaller than the membrane openings flow through.
3) Freeze Crystallization
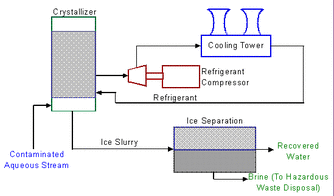
A Typical Freeze Crystallization Unit
Freeze crystallization processes removes purified solvent from solution as frozen crystals.
- Slowly freezing a contaminated solution forms an ice slurry.
- Water freezes at O degreesC - contaminant requires even lower temperatures.
- The less dense water ice crystals form on the surface & the contaminants are concentrated in the remaining solution.
- The ice crystals are then separated, washed and melted to yield a nearly pure water stream.
4) Membrane Pervaporation/Reverse Osmosis
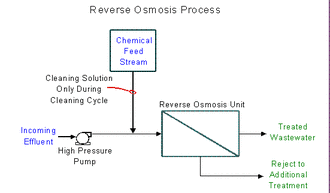
Typical Reverse Osmosis Process
Uses permeable membranes to separate contaminant from solution:
4b) Reverse Osmosis
Reverse osmosis in a modified version of a process called membrane pervaporation process.
- The membranes preferentially adsorb volatile organic compounds (VOCs) from contaminated water.
- Contaminated water first passes through a heat exchanger - to raise water temperature.
- The heated water then enters the pervaporation module (containing the membranes).
- Treated water exits the module, while the organic vapours go to a condenser to return to a liquid.
- The condensed organic materials are now only a fraction of the initial waste-water volume, thus disposed of at large cost savings.
4b) Reverse Osmosis
Reverse osmosis in a modified version of a process called membrane pervaporation process.
- Here, water is forced through the pervaporation module under pressure.
- The module (containing the membranes) allows cleaned water to pass through.
- The water (containing the contaminants) is blocked & recirculated back to the pervaporation module for further treatment - here organic vapours/permeate are extracted by vacuum, condensed and vented downstream of the condenser (minimizing air releases).
Limitations of Separation:
- Freeze crystallization is limited only to aqueous waste streams.
- The presence of oil and grease contaminants may decreasing flow rate through process.
- Distillation and freeze crystallization systems require adequate space (comparatively tall units which cover large areas).
- Explosives that also decompose/polymerize at the operating temperature are not desirable in distillation.
- In freeze crystallization, feed stream must be dilute enough to accomplish significant remediation before the crystalliser solution becomes too concentrated.
