SOIL, SEDIMENT AND SLUDGE: EX-SITU CHEMICAL/PHYSICAL TREATMENT
Chemical Reduction/Oxidation
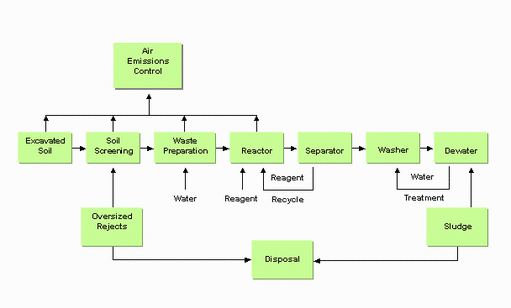
Typical Chemical Reduction/Oxidation Process
Process:
Reduction/oxidation (Redox) reactions use oxidising agents to chemically convert hazardous contaminants to nonhazardous or less toxic compounds that are more stable, less mobile, and/or inert.
Redox reactions involve the transfer of electrons from one compound to another (One reactant is oxidised (loses electrons) and one is reduced (gains electrons)).
The most commonly used oxidising agents are ozone, hydrogen peroxide, hypochlorites, chlorine, and chlorine dioxide.
It is a short- to medium-term technology.
Uses:
Limitations:
Cost:
Approx. between $190-$660 per cubic metre.
Reduction/oxidation (Redox) reactions use oxidising agents to chemically convert hazardous contaminants to nonhazardous or less toxic compounds that are more stable, less mobile, and/or inert.
Redox reactions involve the transfer of electrons from one compound to another (One reactant is oxidised (loses electrons) and one is reduced (gains electrons)).
The most commonly used oxidising agents are ozone, hydrogen peroxide, hypochlorites, chlorine, and chlorine dioxide.
It is a short- to medium-term technology.
Uses:
- Target treatment group is inorganics.
- Also used but less effective for non-halogenated VOCs and SVOCs, fuel hydrocarbons & pesticides.
Limitations:
- Incomplete oxidation may occur depending upon the contaminants & oxidizing agent used (forming intermediate & possibly hazardous contaminants).
- Not cost-effective for high contaminant concentrations (large quantity of oxidizing agent required).
- Presence of oil and grease in the media reduces efficiency.
Cost:
Approx. between $190-$660 per cubic metre.
