SOIL, SEDIMENT AND SLUDGE: IN-SITU CHEMICAL/PHYSICAL TREATMENT
Chemical Oxidation
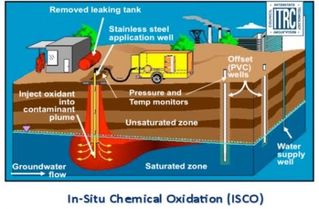
Oxidation chemically converts hazardous contaminants to non-hazardous/less toxic compounds that are more stable, less mobile, and usually inert.
-Oxidant delivery systems often employ vertical or horizontal injection wells and air-sparging wells to rapidly move the oxidant into the subsurface.
-The oxidizing agents most commonly used are: ozone, hydrogen peroxide, hypochlorites, chlorine, and chlorine dioxide.
-Note: Must consider the contaminants and site conditions in selecting the most successful oxidant.
Ozone, Hydrogen Peroxide and Permanganate result in:
-rapid & complete chemical destruction of many toxic organic chemicals, unsaturated aliphatic (e.g TCE) and aromatic compounds (e.g. benzene)
-partial degradation of other organics (aiding their further treatment)
1) Ozone addition
- Ozone gas can oxidize contaminants directly or through the formation of hydroxyl radicals (OH-).
- Extremely rapid oxidation reaction if very acidic pH (2-4).
- Due to ozone’s high reactivity & instability, O3 is produced - may lead to beneficial oxygenation & bio-stimulation.
- Due to ozone’s high reactivity & instability, O3 is produced - may lead to beneficial oxygenation & bio-stimulation.
- Oxidation using liquid H2O2 in the presence of ferrous iron (Fe+2) produces a reagent which yields free hydroxyl radicals (OH-).
- Rapid break down of many organic compounds
- Most effective under very acidic pH (2-4), but ineffective under alkaline conditions.
- Permanganate is typically provided as liquid or solid KMnO4 to destruct the contaminants by direct electron transfer or free radical advanced oxidation.
- Much slower rate than the previous two reactions.
Advantages/Uses:
- Effective treatment for many bio-recalcitrant organics and subsurface environments.
- Relatively simple & readily available equipment.
Disadvantages/Limitations:
- Some contaminants are resistant to oxidation.
- All three oxidation reactions may decrease the soil pH.
- Permanganate (more stable & more persistent in ground) - can migrate by diffusion.
- May cause reduced permeability; mobilisation of sorbed metals; toxic byproducts, heat and gas.
- The rate is dependent on the chemical, soil conditions, pH, temperature & concentration of the oxidant.
- Must ensure safe handling of chemicals at all times.
- No cost information currently available.
