GROUNDWATER AND LEACHATE: IN-SITU CHEMICAL/PHYSICAL TREATMENT
Chemical Oxidation
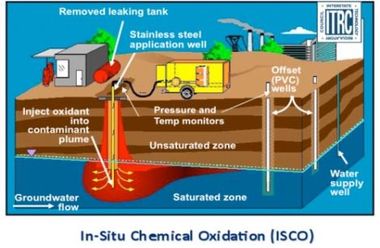
Oxidation chemically converts hazardous contaminants to non-hazardous/less toxic compounds that are more stable, less mobile, and usually inert.
-Oxidant delivery systems often employ vertical or horizontal injection wells and air-sparging wells to rapidly move the oxidant into the subsurface.
-The oxidizing agents most commonly used are: ozone, hydrogen peroxide, hypochlorites, chlorine, and chlorine dioxide.
-Note: Must consider the contaminants of concern (COCs) and site conditions in selecting the most successful oxidant.
Ozone, Hydrogen Peroxide and Permanganate have been known to result in:
-rapid & complete chemical destruction of many toxic organic chemicals
-rapid & >90% efficient treatment of unsaturated aliphatic (e.g TCE) and aromatic compounds (e.g. benzene)
-partial degradation of other organics (aiding their further treatment).
1) Ozone addition
- Ozone gas can oxidize contaminants directly or through the formation of hydroxyl radicals (OH-).
- The oxidation reaction proceeds with extremely fast, pseudo first order kinetics: most effective under very acidic pH (2-4)
- Due to ozone’s high reactivity and instability, O3 is produced onsite, requiring closely spaced air sparging wells.
- In situ decomposition of the ozone can lead to beneficial oxygenation and bio-stimulation.
2) Hydrogen Peroxide (H2O2)
- Oxidation using liquid H2O2 in the presence of ferrous iron (Fe+2) produces a reagent which yields free hydroxyl radicals (OH-).
- These can rapidly degrade a variety of organic compounds and follow second-order kinetics: most effective under very acidic pH (2-4), but ineffective under alkaline conditions.
3) Permanganate
- Permanganate is typically provided as liquid or solid KMnO4 (also available in Na, Ca, or Mg salts) to cause much more complex reactions due to its multiple valence states and mineral forms.
- The reactions follow second order kinetics but are at a much slower rate than the previous two reactions: now effective over a pH range of 3.5 to 12
- Depending on pH, the reaction can include destruction by direct electron transfer or free radical advanced oxidation.
Advantages/Uses:
- Effective treatment (mass reduction) in source and plume areas.
- Rapid and extensive reactions for many bio-recalcitrant organics and subsurface environments.
- Can be tailored to a specific site and implemented with relatively simple, readily available equipment.
Disadvantages/Limitations:
- Some contaminants are resistant to oxidation.
- All three oxidation reactions can decrease the pH if system not buffered effectively.
- Permanganate is relatively more stable and more persistent in the subsurface - can migrate by diffusion.
- Oxidation-induced effects include: reduced permeability; mobilization of redox-sensitive and exchangeable sorbed metals; possible formation of toxic byproducts & evolution of heat and gas.
- The rate and extent of degradation are dependent on the chemical itself, soil conditions, pH, temperature & concentration of the oxidant.
- Must ensure safe handling of chemicals at all times.
- No cost information currently available.
