Off-gas/Waste Vapour/Hazardous Volatile Treatment
Oxidation
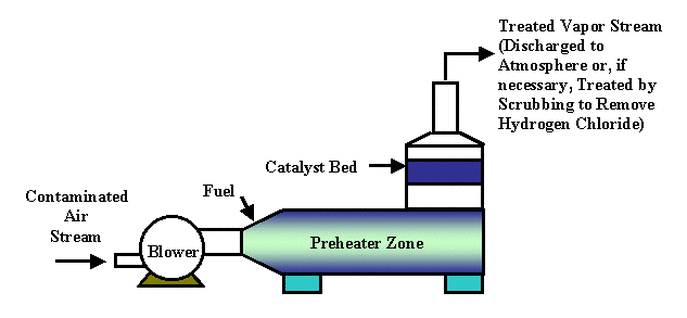
Typical Oxidation Diagram
Oxidation equipment (thermal or catalytic) is used for destroying contaminants in the exhaust gas from air strippers & SVE systems.
Thermal oxidation units typically include single chamber, refractory-lined oxidizers with a propane or natural gas burner and stack (lightweight ceramics used for the refractory lining).
Uses:
- To treat contaminants in the exhaust gas from air strippers and SVE systems.
- Target contaminant groups for oxidation: non-halogenated VOCs and SVOCs, and fuel hydrocarbons.
- Organic contaminants are destroyed at high temperatures (1000°C) in heat-exchanger.
- Trace organics (VOCs) in contaminated air streams are destroyed at lower temperatures (~450 °C) by the use of a catalyst.
- Metal catalysts effectively destroy halogenated & chlorinated hydrocarbons (TCE, TCA, methylene chloride, and 1,1-DCA ).
Treatment Process:
- The contaminated air is directly preheated (using natural gas/propane or electrically) to 310-370 °C to initiate the catalytic oxidation of the VOCs.
- The preheated (VOC-laden) air is passed through a bed of solid catalysts - VOCs are rapidly oxidized.
1) For Gasoline contaminant:
- Heat exchanger efficiencies are limited to 25 to 35%
- Preheat temperatures maintained < 180 °C - reduce possibility of ignition
- Flame arrestors are installed - between vapour source & thermal oxidizer.
- Burner capacity (in combustion chamber): between 0.5- 2 million Btus/hr.
- Operating temperature: 760-870 °C.
2) To treat VOCs in air streams, Catalytic oxidation is applied. The catalyst is added:
- to accelerate the rate of oxidation - it absorbs the oxygen and contaminant on its' surface (where they react to form CO2, water, and HCl(gas)).
- as it enables oxidation to occur at much lower temperatures - VOCs thermally destroyed between 320° to 540° C
The 4 Oxidation Types:
a) Catalytic Oxidation
- Catalyst systems use metal oxides or noble metals to oxidize VOCs, e.g. nickel oxide, copper oxide, manganese dioxide, platinum or palladium.
b) Internal Combustion Engine Oxidation
- Use of the organic contaminants in air as fuel to be burned in an internal combustion engine.
- When the concentration of organics is too low, auxiliary fuel is added to enhance the oxidation.
c) Thermal Oxidation
- Thermal or catalytic oxidation process is enhanced by using an air-to-air heat exchanger to transfer heat from the exhaust gases to the incoming contaminated air - reduces fuel costs as recovers approx. 50% of the heat.
- In UV oxidation, the chemical bonds of the organic contaminants in the air are broken due to the UV light.
- Products of photo-degradation vary but complete degradation of the organic contaminant (to CO2, H2O, etc.) is unlikely.
Note:
-Thermal oxidation is effective for site remediation - of increasing use for treatment of air stripper and vacuum extraction gas-phase emissions.
-Catalytic oxidation is also well established & is continuously progressing to increase the destruction efficiency.
Limitations:
- The presence of chlorinated hydrocarbons,some heavy metals (e.g., lead), sulfur or halogenated compounds may poison a particular catalyst - deactivates catalyst, thus requiring replacement.
- Destruction of halogenated compounds requires special catalysts, special materials or construction, and the addition of a flue gas scrubber to reduce acid gas emissions.
- Influent gas concentrations must be < 25% of the explosive limit for catalytic and thermal oxidation.
Cost:
The cost depends on the flow rate, fuel costs & the quantity, concentration, and type of contaminant.
- At low flow rates or very high VOC concentrations: Catalytic oxidation is more expensive than thermal oxidation.
- At high flow rates or at moderate VOC concentrations: Catalytic oxidation is less expensive than thermal oxidation.
